Breadcrumb
- Home
- Diagnostics
Diagnostics
Diagnostic Information
With leishmaniasis endemic in 88 countries, emergence in the United States, and the zoonotic nature of the disease, identification of infected canines remains of utmost importance so transmission can be minimized. Infected canines are the number one reservoir for human infection. One of the diagnostic tests used in this lab determines whether an animal has been infected with Leishmania infantum through antibody production. An enzyme-linked immunosorbent assay, or ELISA, is a test that measures if the serum sample contains antibodies specific for a disease.
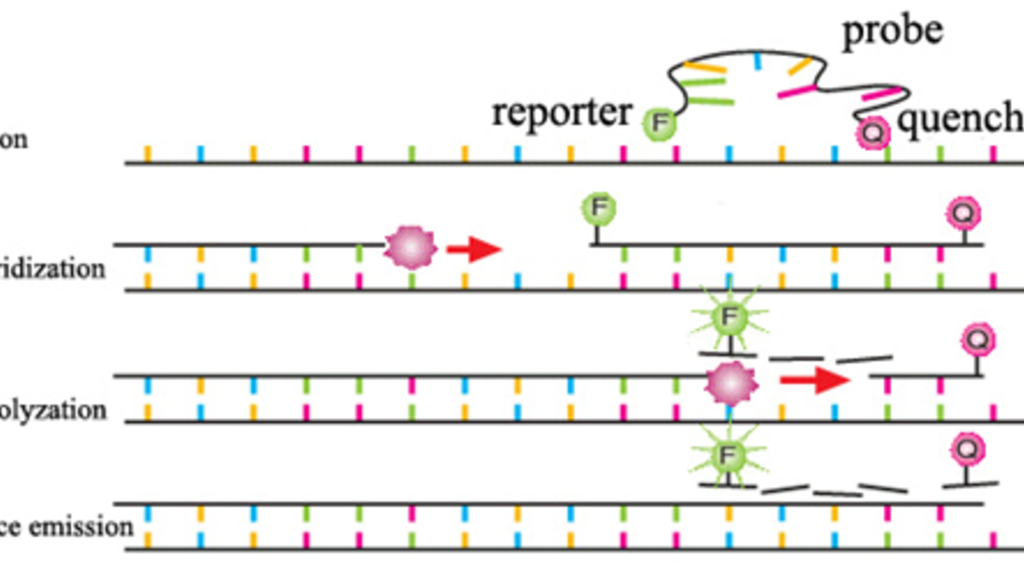
A second diagnostic test ascertains the presence of Leishmania infantum infection through kinetoplast-specific quantitative PCR (qPCR), in which a target sequence of the parasitic kDNA is tagged with a fluorescent probe and replicated repeatedly. This results in exponential amplification, allowing quantification of the original amount of the target sequence. Other tests for leishmaniasis and the L. infantum parasite exist while there is still no reliable vaccine for the disease.
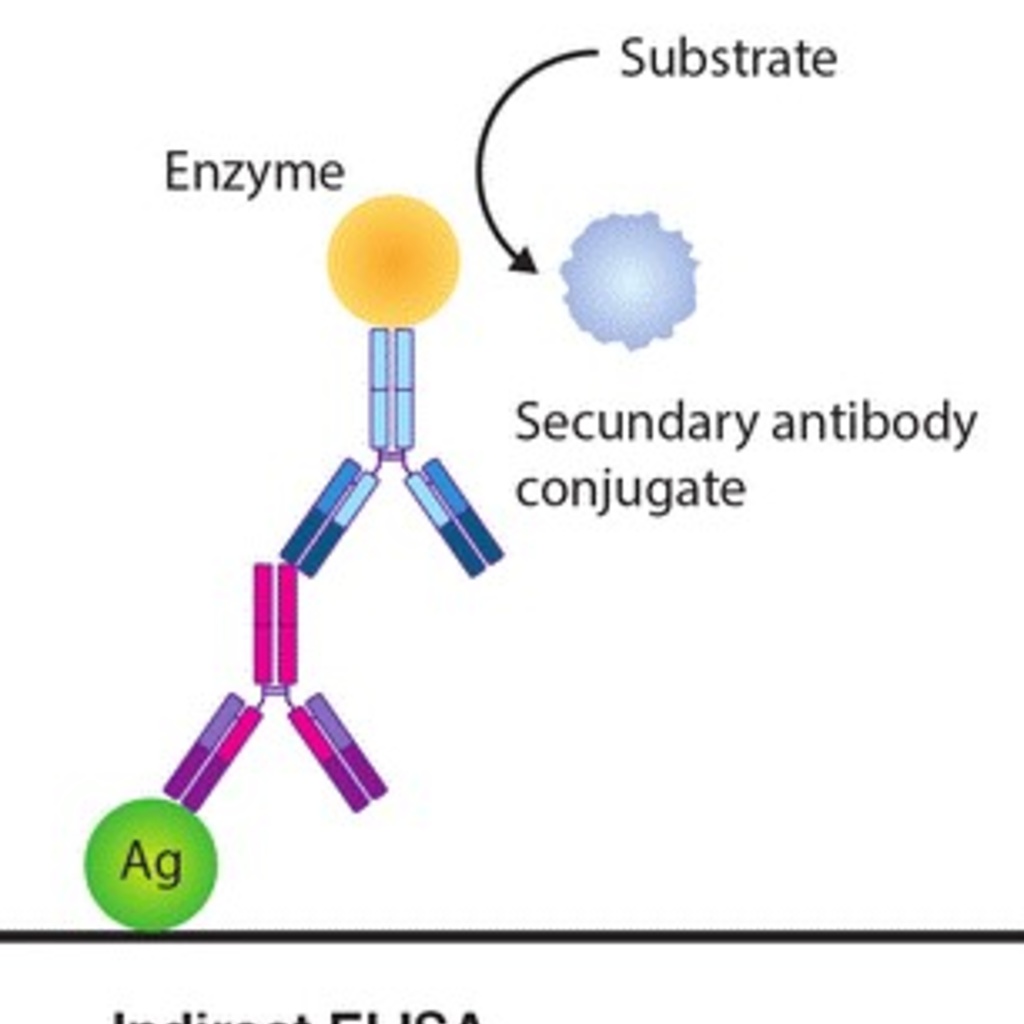
Diagram of ELISA